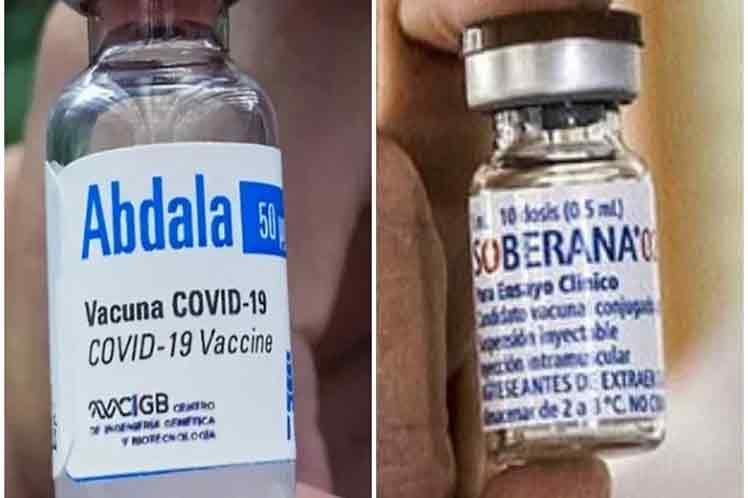
Two vaccine candidates – Soberana 02 and Abdala – are now in the 3rd phase of clinical trials, with ‘efficacy data expected in June,’ stated Rolando Perez, director of Science and Innovation of Cuba’s Biotechnological and Pharmaceutical Industries Group (BioCubaFarma).
Indeed, positive data on the safety and immunogenicity of those preparations were obtained in previous clinical trials, the scientist commented.
Perez defended Cuba’s decision to produce its own drugs to control the pandemic based on the capabilities of the national biotechnology industry.
The immunization program will cover, in a staggered manner, at-risk and vulnerable groups, adults and, finally, the pediatric population, the executive explained.
The expert ratified that the country plans to complete the vaccination of the entire population before the end of 2021, and called attention to the shortage of supplies of raw materials and primary packaging material for the pharmaceutical industry.
This situation is further aggravated in Cuba due to the US economic and financial blockade for over 60 years ago, and which was tightened during the Trump administration (2016-2021).
rly/omr/ga/ro