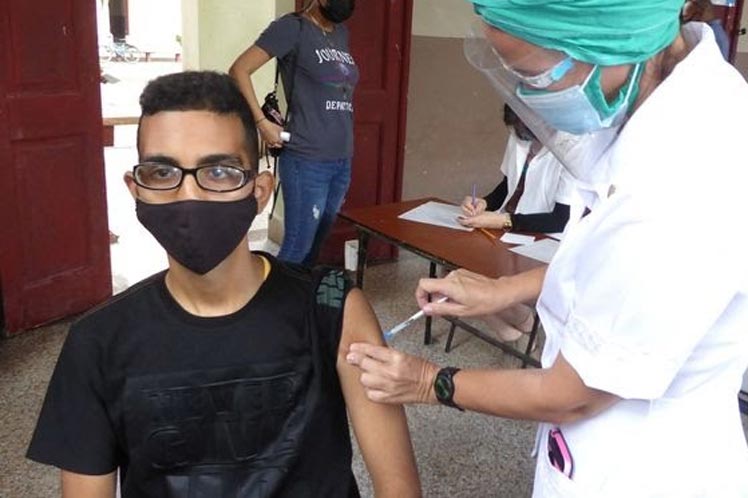
‘The future will be vaccinated’ with the authorization of the Center for State Control of Medicines, Equipment and Medical Devices (Cecmed) for the emergency use of Soberana 02 in ages from 2 to 18, the president wrote on his official Twitter account.
‘Thanks to the children and teenagers who put their shoulders to the trial. Cuba will also win this battle for life,’ he added.
Cecmed said the day before that the vaccine against SARS-CoV-2 proved that it ‘meets the requirements in terms of quality, safety and immune genicity for this population group’.
The nationally produced vaccine was administered with a two-dose scheme in the pediatric population.
The trial with minors, compared with the results of a similar trial in the population group aged between 19 and 80 years, proved to be effective, the entity specified.
The results obtained in the trial in the pediatric population were superior in all immunological variables compared to the adult population aged 19 to 80 years and similar compared to the subgroup of young adults aged 19 to 29 years,’ the regulating institution stated.
mh/abo/ga/ebr