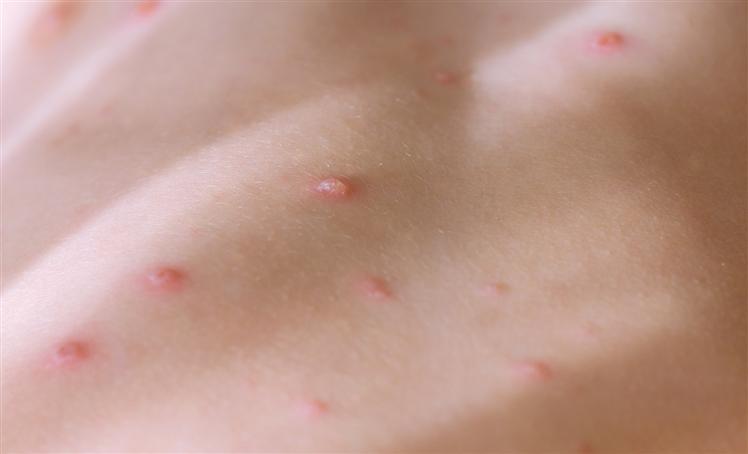
“ANVISA passed to registration exemption for the Health Ministry to import and use Jynneos/Imvanez vaccine nationwide, in order to immunize Brazilians against monkeypox,” the regulatory body informed in a note.
Such permit applies to Jynneos (United States) or Imvanex (Europe) vaccines, which are the same drug but with different names. It was developed by the Bavarian Nordic company and is manufactured in both Denmark and Germany.
To grant approvals, ANVISA analyzed data from the European Medicines Agency and the American Agency.
The temporary and exceptional dispensation applies only to the Health Ministry and will be valid for six months, provided it is not expressly revoked by the regulatory agency.
According to the World Health Organization (WHO), this immunizer is already approved to be used in the United States, Canada and the European Union.
In early August, Health Minister Marcelo Queiroga stated his country would receive Tecovirimat antiviral to fight off the outbreak.
The drug was released in the United States and is manufactured by the multinational Catalent Pharma Solutions. It comes in capsule form, is administered orally and is recommended for treating diseases caused by Orthopoxvirus in adults, adolescents and children weighing at least 13 kg.
Research published in the scientific journal The Lancet Infectious Diseases pointed out that Tecovirimat antiviral showed promise in reducing the length of symptoms and the time in which patients are able to infect other people.
Brazil is the world´s third country reporting most monkeypox cases, preceded by the United States (15,877) and Spain (6,284).
pll/ro/ocs