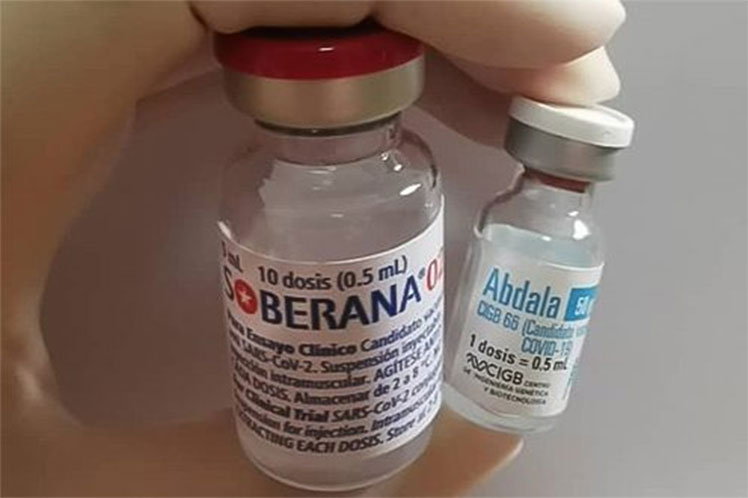
The first Latin American country to present a proposal to counteract the pandemic that has hit the world since 2020, Cuba has administered at least one shot of Soberana or Abdala vaccine candidates to 1,115,335 people until almost halfway through 2021.
Soberana 02, a vaccine candidate developed by the Finlay Vaccines Institute (IFV), was also the first in the region to enter Phase 3 of clinical trials with 44,000 volunteers from Havana, and at this stage, it reinforced the two-dose schedule with another vaccine created at the same institution: Soberana Plus.
Meanwhile, Abdala, named after a poem by Cuban National Hero Jose Marti and conceived at the Genetic Engineering and Biotechnology Center(CIGB), could complete its three-dose schedule in the third phase of clinical trials with the involvement of 48,290 individuals in the eastern provinces of Santiago de Cuba, Granma and Guantanamo.
Both vaccines proved their safety and immunogenicity in each phase, as they did not cause serious adverse events after vaccination and managed to increase the necessary antibody titers, as well as to raise individuals’ immune response, according to their developers.
In all phases of clinical trials, 172,233 Cubans received the planned shots.
For these reasons, health authorities were authorized to initiate, with the vaccine candidates Soberana 02 and Abdala, an intervention study that includes sectors with high risk of contagion, disease and pandemic spread, exposed in real situations of community transmission.
With this evaluation, whose objective is to demonstrate the efficacy, effectiveness and impact of the candidate on the individual to whom it is administered and to know if it protects those who are directly linked to it and have not been immunized, the vaccination candidates have been administered 330,726 people.
jg/aph/oda/cdg